Project
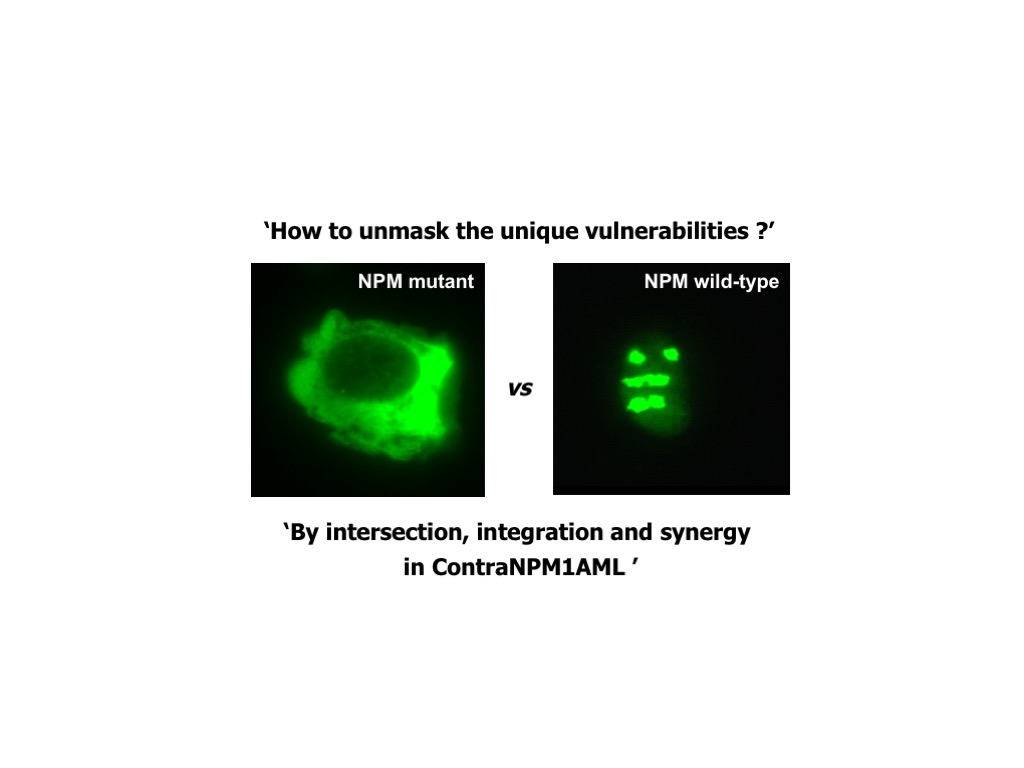
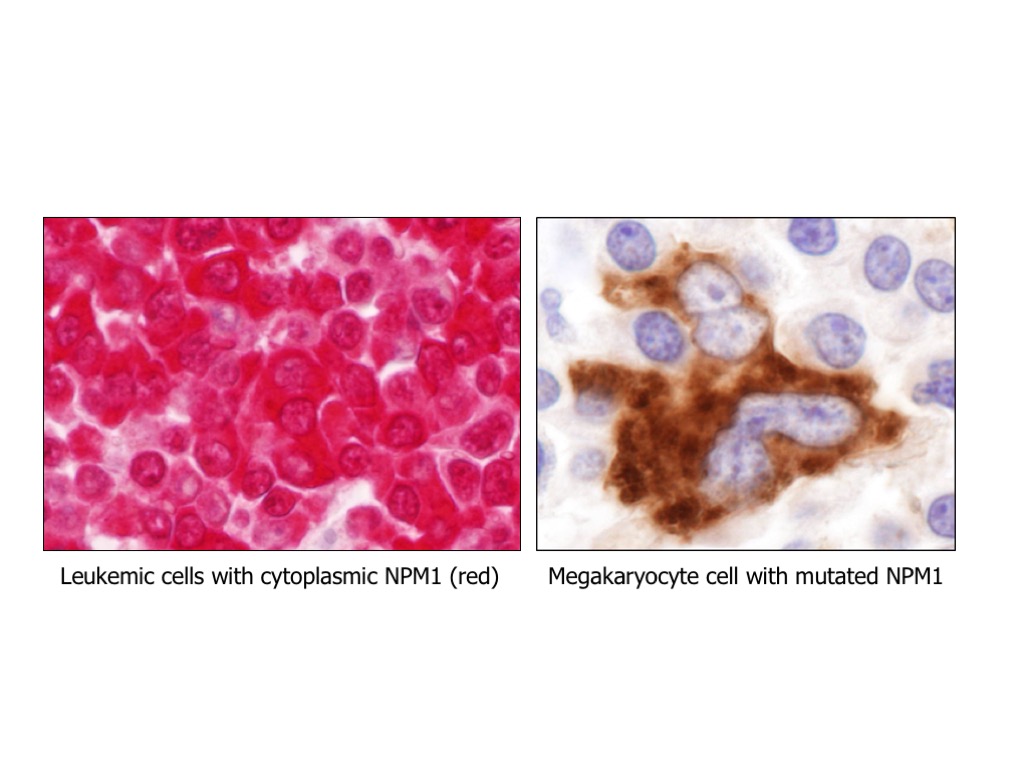
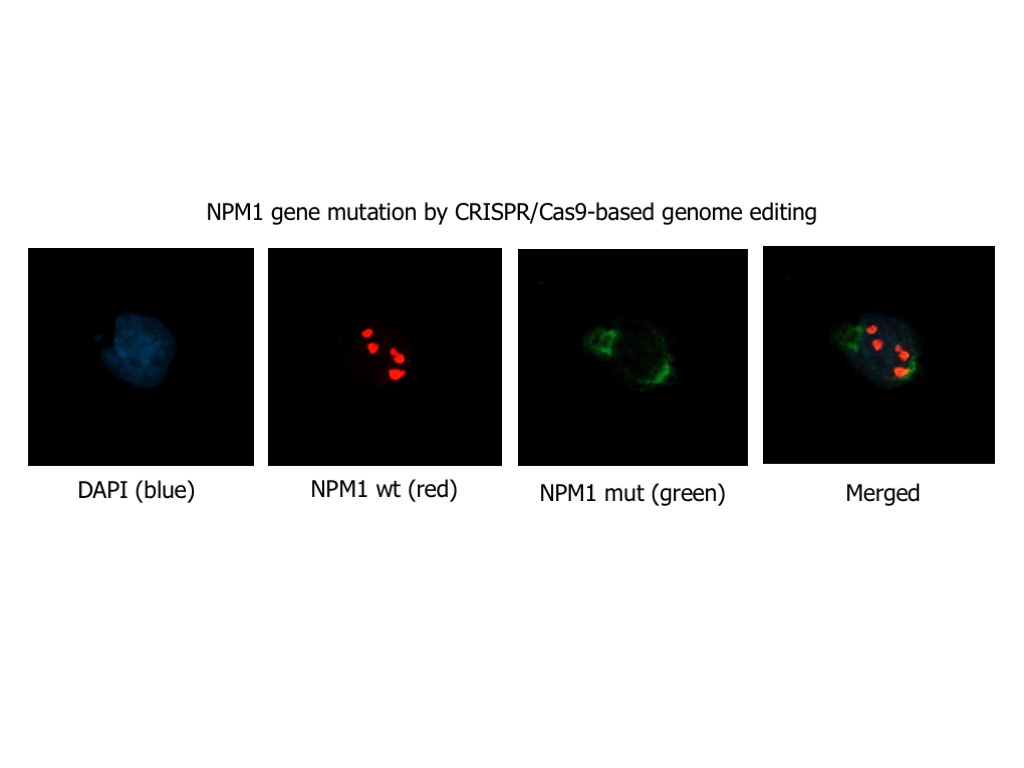
In the wide genomic landscape of acute myeloid leukemia (AML), a major breakthrough was the discovery in 2005, by the research group lead by Prof. Brunangelo Falini at Perugia University, of the NPM1 gene mutations as the most frequent genetic lesion in AML, accounting for about one-third of all adult AML. Today, NPM1-mutated AML is a new entity in the World Organization of Health (WHO) classification of myeloid neoplasms, and NPM1 mutations are one of the most important biomarkers currently used in the clinic for both prognostic stratification of patients and treatment decisions, and monitoring of minimal residual disease (MRD). Nevertheless, NPM1-mutated AML remains a medical need, since still many patients, particularly older patients, succumb to their disease.
At present, there are only few examples of molecularly targeted therapies for AML and only one, i.e. acute promyelocytic leukemia (APL), where the therapy, i.e. the differentiating agent all-trans-retinoic acid (ATRA) and arsenic trioxide (ATO), targets levels and function of the disease-defining genetic lesion, i.e. the PML-RARa oncogenic fusion protein. This has dramatically changed the outcome of patients affected by APL, allowing cure rates above 90%.
It is my believe that to find a cure for AML there is the need to concentrate into the specific genetic subtypes with genetic lesions defining ‘diseases’, that means entities with individual clinical and biological features, well recognized in the WHO classification of tumors, like NPM1-mutated AML is.
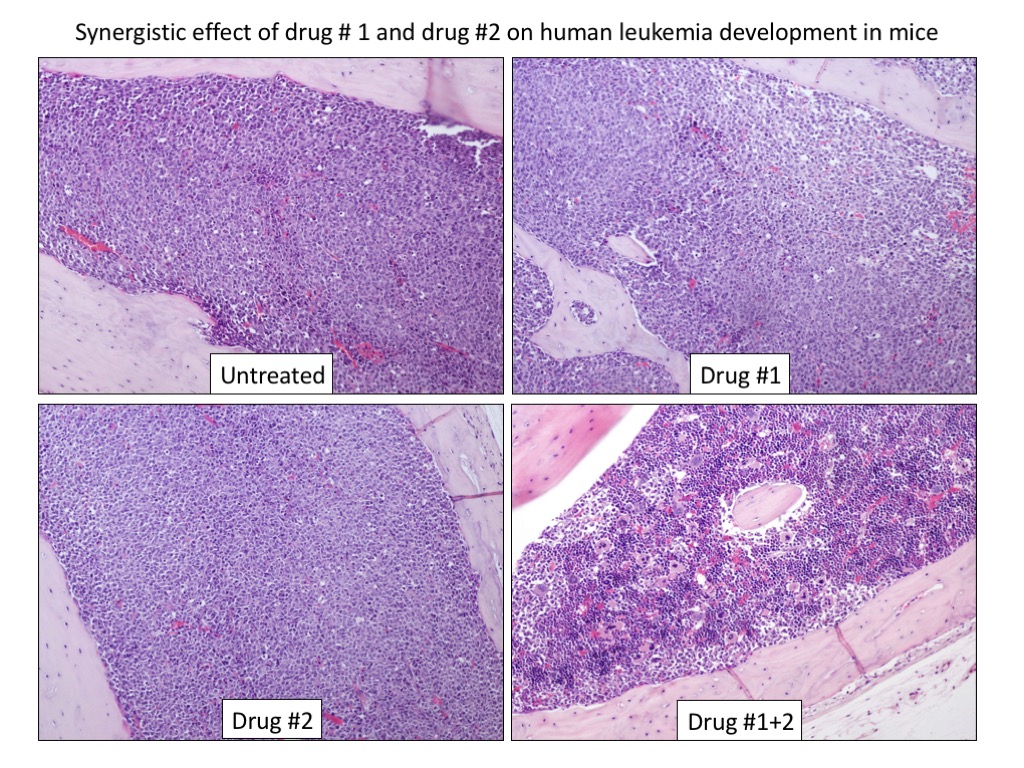
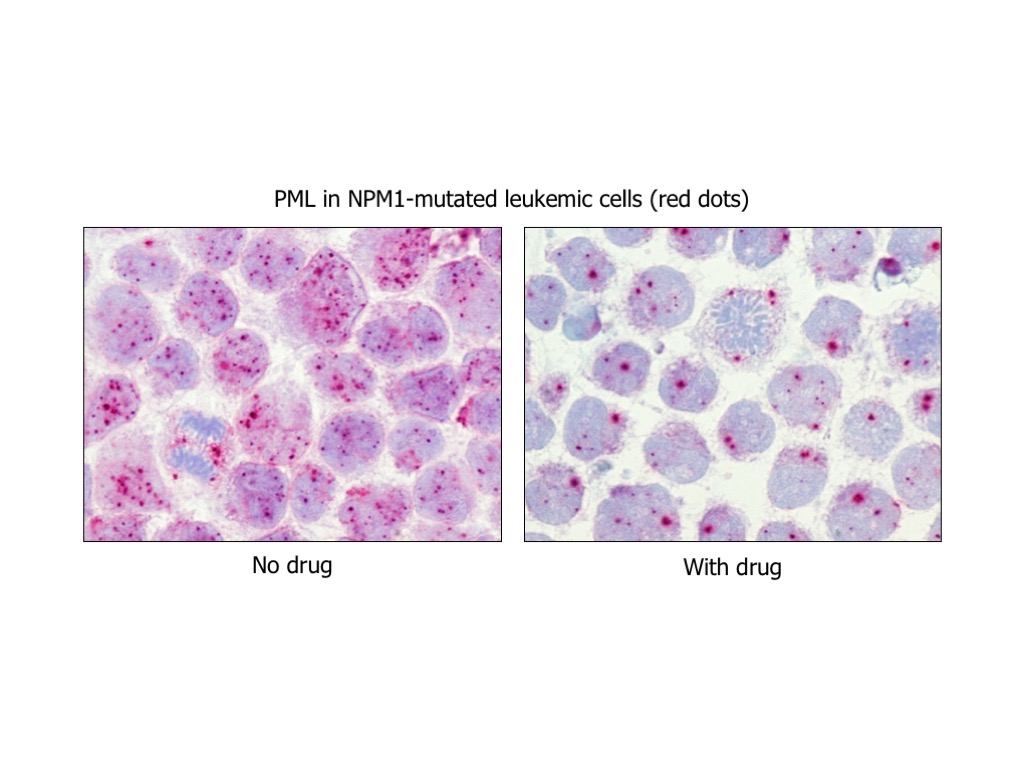
Focusing on this specific genetic subtype, based on our previous characterization studies and preliminary observations, we will use either hypothesis-driven or screening-based approaches to either hit directly NPM1 mutant or identify disease-specific vulnerabilities (‘synthetic lethal’ interactions with NPM1 mutation) in order to: i) dissect leukemogenesis pathways; ii) identify/exploit sensitive pathways and chemical compounds/drugs that kill specifically AML cells harbouring NPM1 mutations; iii) design and develop clinical trials to prove the safety and efficacy of promising new treatments and make them readily available to patients.
Publications
Brunetti L, Gundry MC, Sorcini D, Guzman AG, Huang YH, Ramabadran R, Gionfriddo I, Mezzasoma F, Milano F, Nabet B, Buckley DL, Kornblau SM, Lin CY, Sportoletti P, Martelli MP, Falini B, Goodell MA. Mutant NPM1 Maintains the Leukemic State through HOX Expression. Cancer Cell. 2018 Sep 10;34(3):499-512.e9. doi: 10.1016/j.ccell.2018.08.005.